ISSN: 1449-1907International Journal of Medical Sciences
Int J Med Sci 2024; 21(5):965-977. doi:10.7150/ijms.93720 This issue Cite
Research Paper
The crucial relationship between miRNA-27 and CSE/H2S, and the mechanism of action of GLP-1 in myocardial hypertrophy
1. State Key Laboratory of Frigid Zone Cardiovascular Diseases (SKLFZCD), Department of Pharmacology (State Key Laboratory-Province Key Laboratories of Biomedicine-Pharmaceutics of China, Key Laboratory of Cardiovascular Research, Ministry of Education), College of Pharmacy, Harbin Medical University, Harbin 150081, China.
2. Research Unit of Noninfectious Chronic Diseases in Frigid Zone (2019RU070), Chinese Academy of Medical Sciences, Harbin 150081, China.
3. Department of Pharmacy, The 2nd Affiliated Hospital of Dalian Medical University, Dalian 116023, China.
Abstract
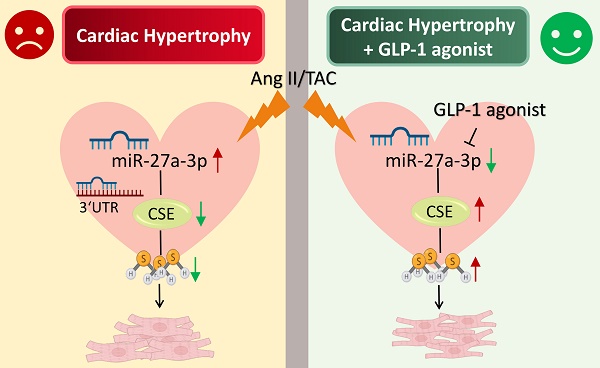
Cardiac hypertrophy is the most prevalent compensatory heart disease that ultimately leads to spontaneous heart failure. Mounting evidence suggests that microRNAs (miRs) and endogenous hydrogen sulfide (H2S) play a crucial role in the regulation of cardiac hypertrophy. In this study, we aimed to investigate whether inhibition of miR-27a could protect against cardiac hypertrophy by modulating H2S signaling. We established a model of cardiac hypertrophy by obtaining hypertrophic tissue from mice subjected to transverse aortic constriction (TAC) and from cells treated with angiotensin-II. Molecular alterations in the myocardium were quantified using quantitative real time PCR (qRT-PCR), Western blotting, and ELISA. Morphological changes were characterized by hematoxylin and eosin (HE) staining and Masson's trichrome staining. Functional myocardial changes were assessed using echocardiography. Our results demonstrated that miR-27a levels were elevated, while H2S levels were reduced in TAC mice and myocardial hypertrophy. Further luciferase and target scan assays confirmed that cystathionine-γ-lyase (CSE) was a direct target of miR-27a and was negatively regulated by it. Notably, enhancement of H2S expression in the heart was observed in mice injected with recombinant adeno-associated virus vector 9 (rAAV9)-anti-miR-27a and in cells transfected with a miR-27a inhibitor during cardiac hypertrophy. However, this effect was abolished by co-transfection with CSE siRNA and the miR-27a inhibitor. Conversely, injecting rAAV9-miR-27a yielded opposite results. Interestingly, our findings demonstrated that glucagon-like peptide-1 (GLP-1) agonists could mitigate myocardial damage by down-regulating miR-27a and up-regulating CSE. In summary, our study suggests that inhibition of miR-27a holds therapeutic promise for the treatment of cardiac hypertrophy by increasing H2S levels. Furthermore, our findings unveil a novel mechanism of GLP-1 agonists involving the miR-27a/H2S pathway in the management of cardiac hypertrophy.
Keywords: microRNA-27a, cardiac hypertrophy, hydrogen sulfide, cystathionine-γ-lyase, glucagon-like peptide-1 agonist
